Physiological impact of continuous positive airway pressure (CPAP) on total cerebral blood flow in healthy awake volunteers (ultrasound study)
Contents
Team
- Celine Odier
- Thiresia Yiallourou
- Raphael Heinzer
- Lorenz Hirt
- José Haba-Rubio
- Nikolaos Stergiopulos
- Bryn Martin
The problem
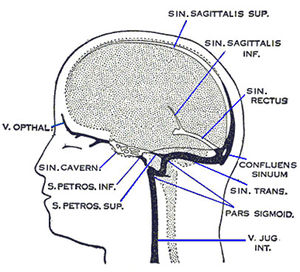
Continuous positive airway pressure (CPAP) is used for the treatment of sleep apnea patients, acting as a pneumatic splint to prevent collapse of the pharyngeal airway by increasing intrathoracic pressure (Bowie, O'Connor et al. 2001). Communication between intrathoracic and intracranial pressure (ICP) through the venous system has been established (Williams 1981; Williams 1981; Heiss, Patronas et al. 1999; Sansur, Heiss et al. 2003) (Figure 1), which are linked with changes in intracranial compliance (Feldman, Robertson et al. 1997) and cardiac output (Feldman, Robertson et al. 1997; Zaramella, Freato et al. 2006). However, the influence CPAP has on cerebral blood flow (CBF) dynamics is not well understood.
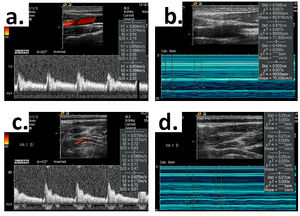
Some studies have quantified CBF velocity using transcranial Doppler ultrasound in the cerebral middle cerebral artery during CPAP (Werner, Kochs et al. 1990; Haring, Hormann et al. 1994; Bowie, O'Connor et al. 2001; Scala, Turkington et al. 2003). However, TCD measurements do not take into account changes in artery diameter which can occur, however minute, resulting from CBF autoregulation. Thus, the precise blood flow rate (ml/s) in the vessels of interest was not obtained, and total CBF was not measured. The proposed study investigates the influence of CPAP on total CBF by measuring velocity, rate, and lumen diameter in the internal carotid and vertebral arteries, and velocity in the middle cerebral arteries using duplex Doppler ultrasound with and without CPAP.
Hypothesis and research objectives
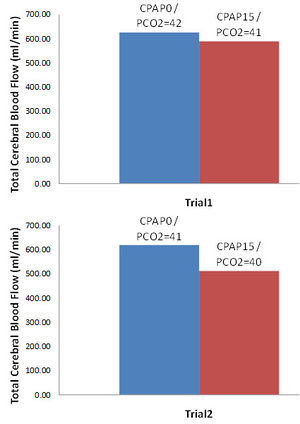
We hypothesize that an increase in CPAP will decrease CBF rate in healthy volunteers. The goal of this research is thus to improve understanding of the influence of CPAP on CBF dynamics in healthy individuals.
Methods and study outline
In vivo Ultrasound measurements. Our approach is to obtain CBF rate measurements before and during CPAP using duplex color Doppler ultrasound on 20 age-matched healthy non smoking volunteers (20-30 yrs.). Each subject will have blood velocity and lumen diameter in the internal carotid and vertebral arteries, and velocity in the middle cerebral arteries, measured with duplex Doppler ultrasound (Soustiel, Glenn et al. 2005). Velocity and diameter measurements will be conducted at least three times at each measurement location. These measurements will be obtained with subjects in the supine position with the head at 30 degrees at atmospheric pressure and after CPAP has been administered for 15 minutes at +15 cm H2O. Transcutaneous carbon dioxide tension PCO2 and oxygen saturation SO2 will be monitored during the entire testing period for each subject. Heart rate and blood pressure will be recorded before and after each test. Pressure test order will be randomized for each subject. Measurement reproducibility will be examined by performing the same measurements three times on three subjects.
Expected results and potential impact
The proposed work will improve understanding of how CBF in healthy volunteers is influenced by CPAP. The test could be further explored to investigate how CPAP influences CBF in patients with sleep apnea and various types of cerebrovascular and craniospinal disorders such as stroke and hydrocephalus.
Preliminary results
Measurement scheduling
http://www.doodle.com/43m7htsurh87b8h2
References
Bowie, R. A., P. J. O'Connor, et al. (2001). "The effect of continuous positive airway pressure on cerebral blood flow velocity in awake volunteers." Anesth Analg 92(2): 415-7.
Feldman, Z., C. S. Robertson, et al. (1997). "Positive end expiratory pressure reduces intracranial compliance in the rabbit." J Neurosurg Anesthesiol 9(2): 175-9.
Haring, H. P., C. Hormann, et al. (1994). "Continuous positive airway pressure breathing increases cerebral blood flow velocity in humans." Anesth Analg 79(5): 883-5.
Harloff, A., C. Strecker, et al. (2006). "Combined measurement of carotid stiffness and intima-media thickness improves prediction of complex aortic plaques in patients with ischemic stroke." Stroke 37(11): 2708-12.
Heiss, J. D., N. Patronas, et al. (1999). "Elucidating the pathophysiology of syringomyelia." J Neurosurg 91(4): 553-62.
Leguy, C. A., E. M. Bosboom, et al. (2009). "Model-based assessment of dynamic arterial blood volume flow from ultrasound measurements." Med Biol Eng Comput 47(6): 641-8.
Lockey, P., G. Poots, et al. (1975). "Theoretical aspects of the attenuation of pressure pulses within cerebrospinal-fluid pathways." Med Biol Eng 13(6): 861-9.
Sansur, C. A., J. D. Heiss, et al. (2003). "Pathophysiology of headache associated with cough in patients with Chiari I malformation." J Neurosurg 98(3): 453-8.
Scala, R., P. M. Turkington, et al. (2003). "Effects of incremental levels of continuous positive airway pressure on cerebral blood flow velocity in healthy adult humans." Clin Sci (Lond) 104(6): 633-9.
Soustiel, J. F., T. C. Glenn, et al. (2005). "Monitoring of cerebral blood flow and metabolism in traumatic brain injury." J Neurotrauma 22(9): 955-65.
Werner, C., E. Kochs, et al. (1990). "[The effect of positive end expiratory pressure on the blood flow velocity in the basal cerebral arteries during general anesthesia]." Anasth Intensivther Notfallmed 25(5): 331-4.
Williams, B. (1981). "Simultaneous cerebral and spinal fluid pressure recordings. 2. Cerebrospinal dissociation with lesions at the foramen magnum." Acta Neurochir (Wien) 59(1-2): 123-42.
Williams, B. (1981). "Simultaneous cerebral and spinal fluid pressure recordings. I. Technique, physiology, and normal results." Acta Neurochir (Wien) 58(3-4): 167-85.
Zaramella, P., F. Freato, et al. (2006). "Does helmet CPAP reduce cerebral blood flow and volume by comparison with Infant Flow driver CPAP in preterm neonates?" Intensive Care Med 32(10): 1613-9.